|
8/29/2023 0 Comments Sigma bond and pi bond examples This carbon, right here, so that carbon has only This carbon over here,Īlso has a double-bond to it, so it's also SP two hybridized, with trigonal planar geometry. Identifying a hybridization state, is to say, "Okay, that carbon has "a double bond to it therefore, it must "be SP two hybridized." And if it's SP two hybridized, we know the geometry around thatĬarbon must be trigonal, planar, with bond anglesĪpproximately 120 degrees. 
Identify the hybridization states, and predict the geometetries for all the atoms in this molecule, except for hydrogen, and so, let's start with this carbon, right here. Understand hybridization states, let's do a couple of examples, and so we're going to So the 3 specifies it is the third carbon in the chain which bears the amino group. The 3 in front says which carbon the amino group is on because a molecule with just aminopropanal could have that amino group on any of the other carbon atoms. As a substituent it gets the name amino. The amine is then what is known as a substituent, or is substituting a hydrogen atom which would otherwise be bonded to a carbon. In organic chemistry aldehydes take priority over amines so this molecule is technically an aldehyde as a class of molecule which is reflected in the suffix of -al. The trick for the name is deciding which one of those functional groups takes priority. The nitrogen part is what is known as an amine while the oxygen part is known as an aldehyde. 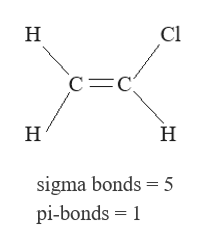
The two important (or unique) parts of the molecule called functional groups are the oxygen and nitrogen parts of it. Because there are only single bonds between the carbon atoms, that makes it 'propan' more specifically as the base name. Three carbons are the backbone of the molecule which give a 'prop' base name. The number of carbons in a row, or a chain, decide what the base name is. If you wonder how we come up with a name like that, here's a brief explanation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
